Disinfection is a core process in drinking water treatment and a critical safeguard for rural drinking water safety. Sodium hypochlorite (NaClO) stands out as a high-efficiency, broad-spectrum disinfectant with excellent disinfection efficacy and residual sterilization capacity, making it the preferred choice for rural and town water supply systems. In recent years, liquid chlorine and chlorine dioxide disinfection have faced application limitations due to raw material safety and handling risks, while sodium hypochlorite disinfection has gradually become the mainstream technical solution for rural water treatment.

This study systematically compares four sodium hypochlorite disinfection application modes—ion membrane electrolysis, conventional diaphragm electrolysis, non-diaphragm electrolysis, and commercial sodium hypochlorite solution—from the perspectives of disinfectant quality, water quality impact, disinfection efficacy, by-product generation, and operating costs. The research aims to screen out the most suitable sodium hypochlorite disinfection technology for rural and town water supply characteristics, providing technical support for improving rural drinking water safety.
Two Main Methods for Obtaining Sodium Hypochlorite for Water Disinfection
Sodium hypochlorite for drinking water disinfection is mainly obtained through two approaches, with significant differences in transportation, storage, and application performance:
1. Commercial sodium hypochlorite solution: Classified as a hazardous chemical, it faces difficulties in purchase and long-distance transportation. High-concentration solutions are prone to decomposition, with poor long-term storage stability, which restricts its popularization in remote rural areas.
2. On-site electrolysis of brine: Using tap water and food-grade salt as raw materials (easily available in rural areas), the solution is prepared and used on demand, ensuring the purity of sodium hypochlorite. The electrolysis generator features simple operation and management, making it more adaptable to the operational characteristics of rural water supply stations with small water volume and simple management.
On-site electrolysis generators are divided into non-diaphragm and diaphragm types based on the presence of a diaphragm between the anode and cathode chambers. Diaphragm materials include traditional nylon membranes and new cation exchange membranes (ion membrane). Chlory brine electrolysis sodium hypochlorite generators cover a full range of models and are the optimal equipment choice for on-site preparation.
Research Methods and Index Measurement Standards
Test Design
The study set up four experimental groups corresponding to the four disinfection modes, and carried out comparative tests from three core dimensions:
1. Disinfectant quality and water quality impact: Compare the effective chlorine concentration stability of sodium hypochlorite solutions produced by the three electrolysis generators and commercial solutions with operation time; add disinfectant with 2mg/L effective chlorine to water samples, and measure pH, TDS, Na⁺ and Cl⁻ concentrations after 30 minutes to analyze water quality changes.
2. Disinfection efficacy and by-product generation: Taking Escherichia coli 8099 (a standard fecal pollution indicator bacterium) as the test object, add 1mg/L effective chlorine disinfectant to the same water body, and detect the bacterial inactivation rate at 1, 3, 5, 7, 10, 20 and 30 minutes; simultaneously measure the generation of typical disinfection by-products (trihalomethane, dichloroacetic acid, trichloroacetic acid) after 30 minutes.
3. Operating cost calculation: Measure the salt and power consumption of the three electrolysis devices during 6 hours of continuous operation, and compare it with the use cost of commercial sodium hypochlorite solution, with the calculation based on market electricity and salt prices.
Index Measurement and Calculation Standards
All test methods strictly follow national and industrial standards to ensure data accuracy and comparability:
- Effective chlorine concentration: Detected by a portable rapid chlorine meter (0-10mg/L), high-concentration solutions (above 10g/L) are tested after gradient dilution, with sampling every 1 hour after the electrolysis device runs stably.
- Chlorate and chlorite: Detected by ion chromatography, each sample is measured 3 times, and the average value is taken as the result.
- Water quality indicators (pH, TDS, Na⁺, Cl⁻): pH and TDS are measured by a pH meter, Na⁺ and Cl⁻ by an ion meter, with 3 parallel measurements for each index.
- Bacterial detection: Escherichia coli 8099 (from China CDC) is cultured at 37℃ for 24 hours after filtration, following the Disinfection Technical Specifications (2002 Edition) and Drinking Water Standard Test Method (GB/T5750-2006).
- Consumption and cost calculation: Salt consumption is calculated based on the change of chloride ion concentration in brine before and after electrolysis; power and salt consumption per unit effective chlorine follow Sodium Hypochlorite Generator (GB12176-1990); operating cost formula: Operating cost = Power consumption × electricity unit price + Salt consumption × salt unit price (unit: yuan/g).
Test Results and Key Analysis
Disinfectant Quality and Water Quality Impact
Effective chlorine concentration is the core index reflecting the oxidation capacity of sodium hypochlorite and the performance of electrolysis generators:
- Concentration stability: The effective chlorine concentration of non-diaphragm and conventional diaphragm electrolysis solutions remains stable with extended operation time; the ion membrane electrolysis solution shows a slow increase in concentration and stabilizes after 5 hours of operation.
- Concentration level: The average effective chlorine concentration of ion membrane electrolysis solution reaches 80.51g/L, 10.75 times that of non-diaphragm electrolysis (7.51g/L), 4.51 times that of conventional diaphragm electrolysis (17.86g/L), and 1.41 times that of commercial solution.
- By-product content: Chlorate concentration in the three electrolysis solutions changes little with operation time; the average chlorate concentration of ion membrane electrolysis solution is only 34.57% of that of conventional diaphragm electrolysis (slightly higher than non-diaphragm electrolysis). Chlorite content in all three electrolysis solutions is at a low level, significantly lower than that of commercial sodium hypochlorite solution.
- Water quality impact: Under the same effective chlorine dosage, the ion membrane electrolysis solution has the smallest impact on raw water pH, TDS and ion concentration, which is due to the cation exchange membrane in its electrolytic cell—only allowing Na⁺ to pass through, preventing Cl⁻ from migrating to the cathode and OH⁻ to the anode, and avoiding side reactions in non-diaphragm cells.
Disinfection Efficacy and Disinfection By-Product Generation
Taking Escherichia coli inactivation rate as the core index of disinfection capacity:
- The bactericidal effects of the three electrolysis-generated sodium hypochlorite solutions are basically equivalent, and all have a significantly higher bactericidal rate than commercial sodium hypochlorite solution at the same contact time.
- In terms of disinfection by-products, the ion membrane electrolysis solution group produces the least trihalomethane, dichloroacetic acid and trichloroacetic acid, which is more in line with the requirements of drinking water safety standards for disinfection by-product control.
Operating Cost Comparison
The salt and power consumption of electrolysis devices are the key factors affecting the operational cost of rural water supply stations (with strict cost control requirements):
- The ion membrane electrolysis sodium hypochlorite generator has the lowest unit salt and power consumption among the three electrolysis devices.
- The average operating cost of ion membrane electrolysis mode is only 0.0078 yuan/m³, which is far lower than the conventional diaphragm electrolysis, non-diaphragm electrolysis and commercial sodium hypochlorite solution modes, with obvious economic advantages.
Core Conclusions of the Study
Based on the comprehensive comparison of multiple indexes, the ion membrane electrolysis sodium hypochlorite disinfection mode is the most suitable technical solution for rural and town drinking water supply, with the following core advantages:
1. High effective chlorine concentration: The concentration is far higher than other electrolysis modes and commercial solutions, and the solution prepared on site has good stability, which meets the disinfection needs of different water volumes in rural areas.
2. Minimal impact on water quality: The cation exchange membrane avoids side reactions, and the impact on raw water pH, ion concentration and TDS is the smallest, ensuring the basic water quality indicators meet the standards.
3. Excellent disinfection efficacy and low by-products: The bactericidal rate is higher than commercial solutions, and the generation of harmful disinfection by-products is the least, which effectively balances disinfection efficacy and water safety.
4. Low operating cost: The unit salt and power consumption is the lowest, and the operating cost is far lower than other modes, which is adapted to the economic operation characteristics of rural water supply systems with small scale and low profit.
5. Strong adaptability: Raw materials (salt and tap water) are easily available in rural areas, the generator is simple to operate and manage, and it is suitable for the actual situation of rural water supply stations with insufficient professional technical personnel.
Chlory Ion Membrane Sodium Hypochlorite Generator
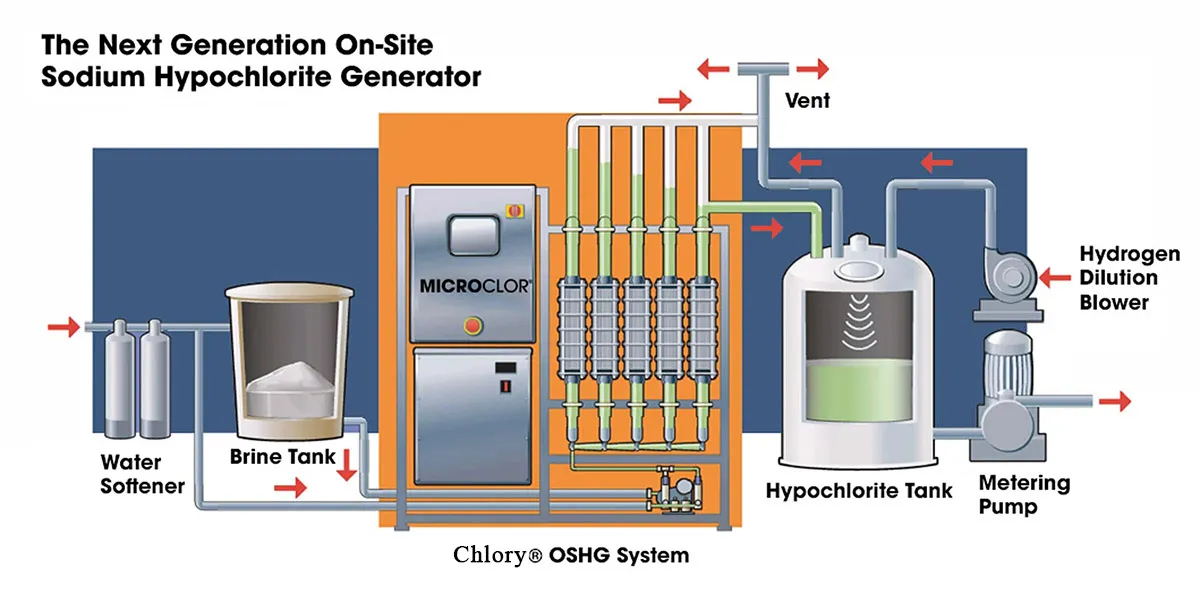
Chlory provides a full range of ion membrane electrolysis sodium hypochlorite generators with a daily output of 100kg to 30 tons, which can be customized according to the water supply scale of rural and town water supply stations (from small villages to large towns). The equipment adopts advanced cation exchange membrane technology, with stable operation, low consumption and simple maintenance, and is the preferred equipment for on-site preparation of sodium hypochlorite in rural drinking water disinfection.







